Product Portfolio
Comprehensive Orthopaedic Care for All
Our portfolio brings together innovative and clinically driven solutions designed to address a broad range of orthopaedic conditions, from joint disorders to bone and cartilage degeneration. Built on robust scientific principles, each solution aims to enhance mobility, support effective recovery and deliver sustained improvements in patient outcomes.

Osteoarthritis Care

Osteoporosis Care
Pain Management / Neuropathic Pain

Autoimmune & Rheumatology
Advancing Therapies. Improving Lives
With Mastro Biologicals, you choose innovation backed by research, expertise and a patient-first mindset. Our solutions are designed to enhance treatment outcomes while ensuring trust, reliability and long-term value
Technology Driven Approach
Patient-Centric & Accessible Care
Highly Experienced Team
Orthopaedic Partnership & Engagement
Comprehensive Orthopaedic Portfolio
Osteoarthritis & Degenerative Joint Disorders
Comprehensive intra-articular and regenerative solutions designed to:
- Restore joint lubrication
- Reduce mechanical degeneration
- Improve mobility and functional performance
- Support long-term cartilage preservation
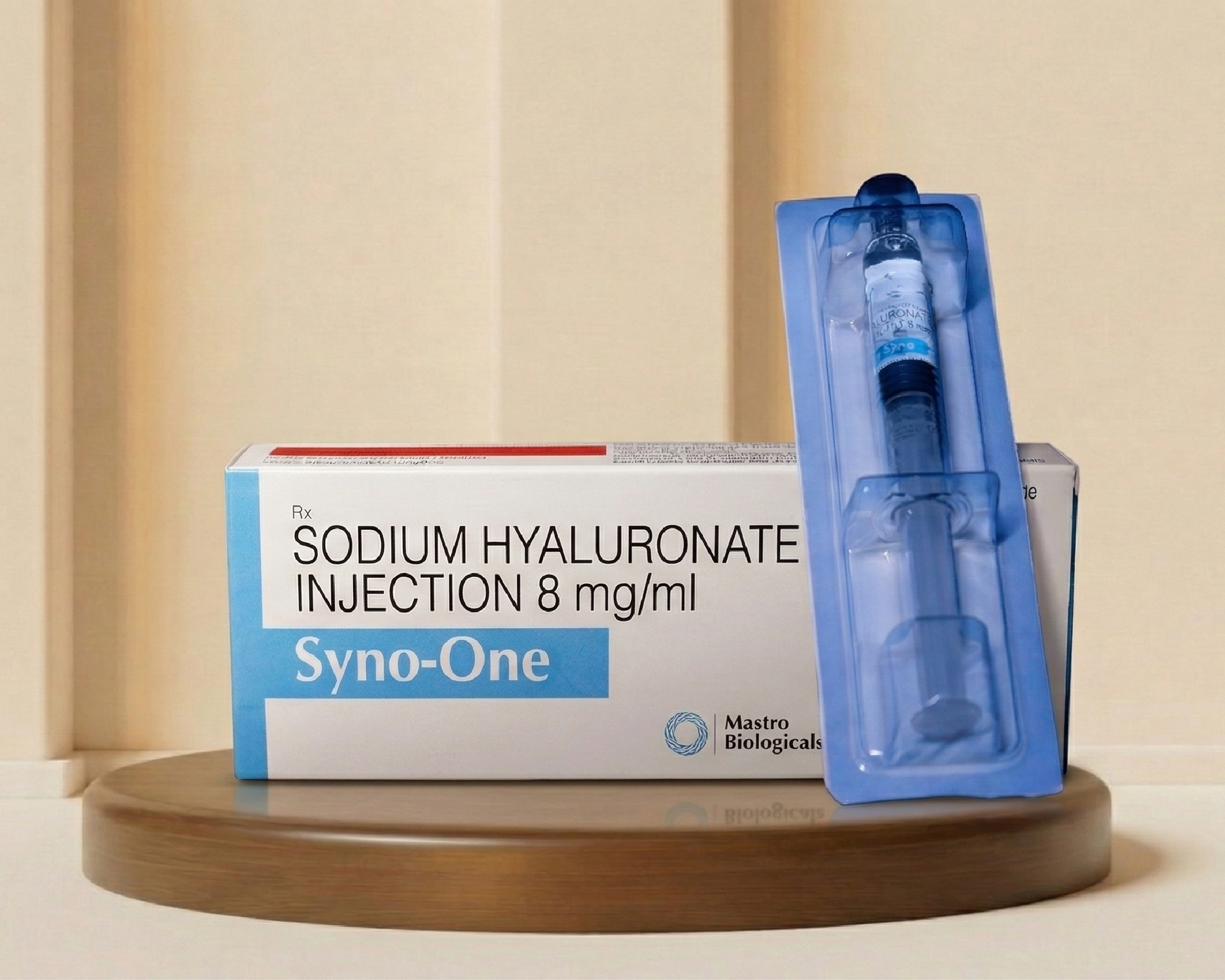
Syno One
Class: Viscosupplement / Intra-articular lubricant
Molecule Type: Non-animal stabilized hyaluronic acid (NASHA)
Scientific Overview
Syno One contains high-molecular-weight sodium hyaluronate designed to restore synovial viscoelasticity in osteoarthritic joints. In OA, endogenous hyaluronic acid becomes depolymerised and loses its natural lubricating and shock-absorbing properties. Exogenous hyaluronate injections replenish viscosity, reduce synovial friction, and modulate intraarticular inflammation.
- Replenishes depleted synovial HA
- Forms a viscoelastic film over cartilage surfaces
- Reduces inflammatory cytokines
- Enhances boundary lubrication
- Improves biomechanical cushioning and shock absorption
- Pain Reduction: Significant reduction in VAS pain scores with improved WOMAC mobility indices
- Lasting Relief: Provides symptom relief lasting 4–6 months. Supports joint homeostasis and may delay need for arthroplasty.
- Broad Indications: Mild to moderate knee OA, hip/shoulder/ankle OA, post-traumatic chondral degeneration, sports-related chondropathy
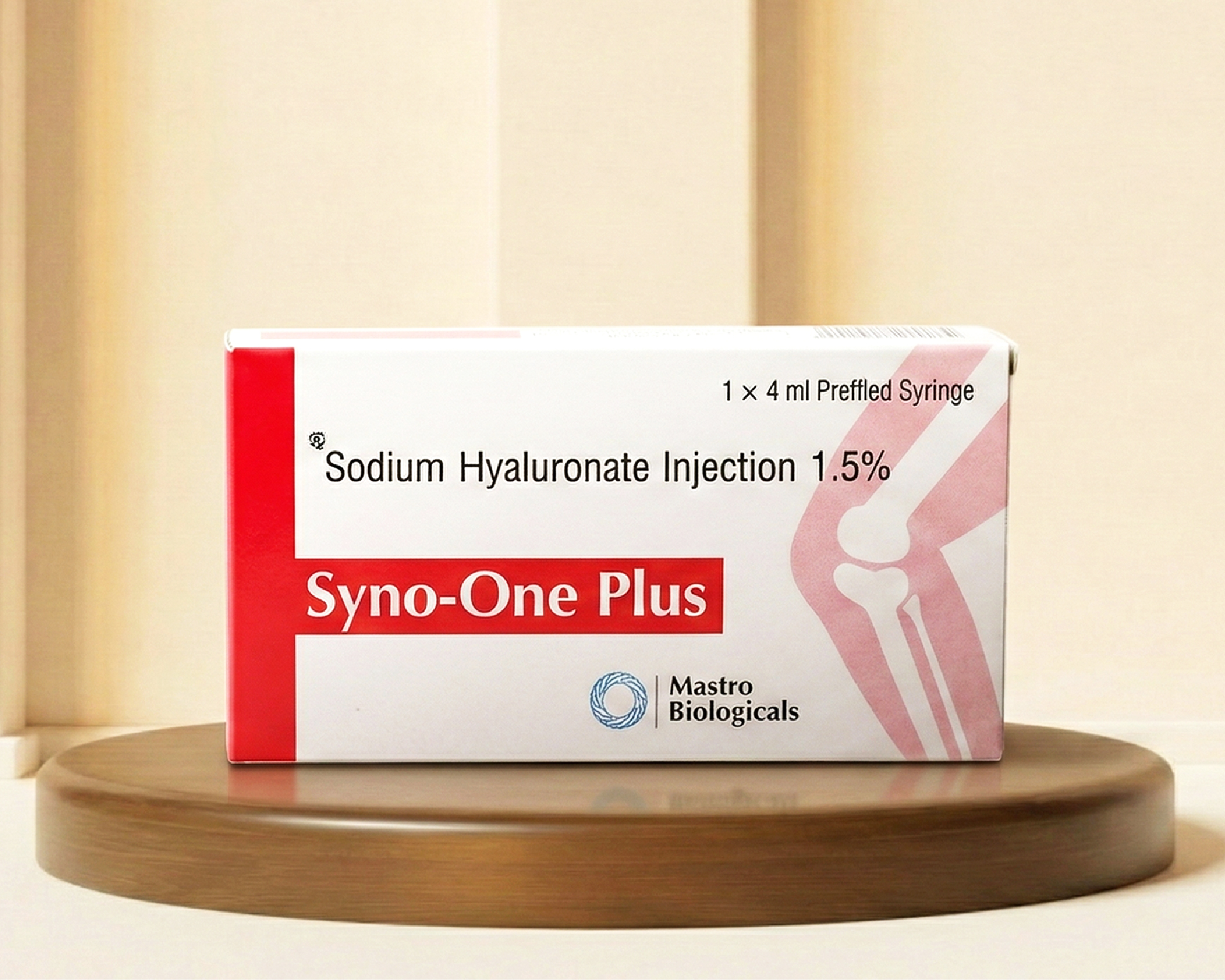
Syno one plus - Sodium Hyaluronate Injection
Class: Viscosupplement / Intra-articular lubricant
Molecule Type: Non-animal stabilized hyaluronic acid (NASHA)
Scientific Overview
Syno One contains high-molecular-weight sodium hyaluronate designed to restore synovial viscoelasticity in osteoarthritic joints. In OA, endogenous hyaluronic acid becomes depolymerised and loses its natural lubricating and shock-absorbing properties. Exogenous hyaluronate injections replenish viscosity, reduce synovial friction, and modulate intraarticular inflammation.
- Replenishes depleted synovial HA
- Forms a viscoelastic film over cartilage surfaces
- Reduces inflammatory cytokines
- Enhances boundary lubrication
- Improves biomechanical cushioning and shock absorption
- Pain Reduction: Significant reduction in VAS pain scores with improved WOMAC mobility indices
- Lasting Relief: Provides symptom relief lasting 4–6 months. Supports joint homeostasis and may delay need for arthroplasty.
- Broad Indications: Mild to moderate knee OA, hip/shoulder/ankle OA, post-traumatic chondral degeneration, sports-related chondropathy
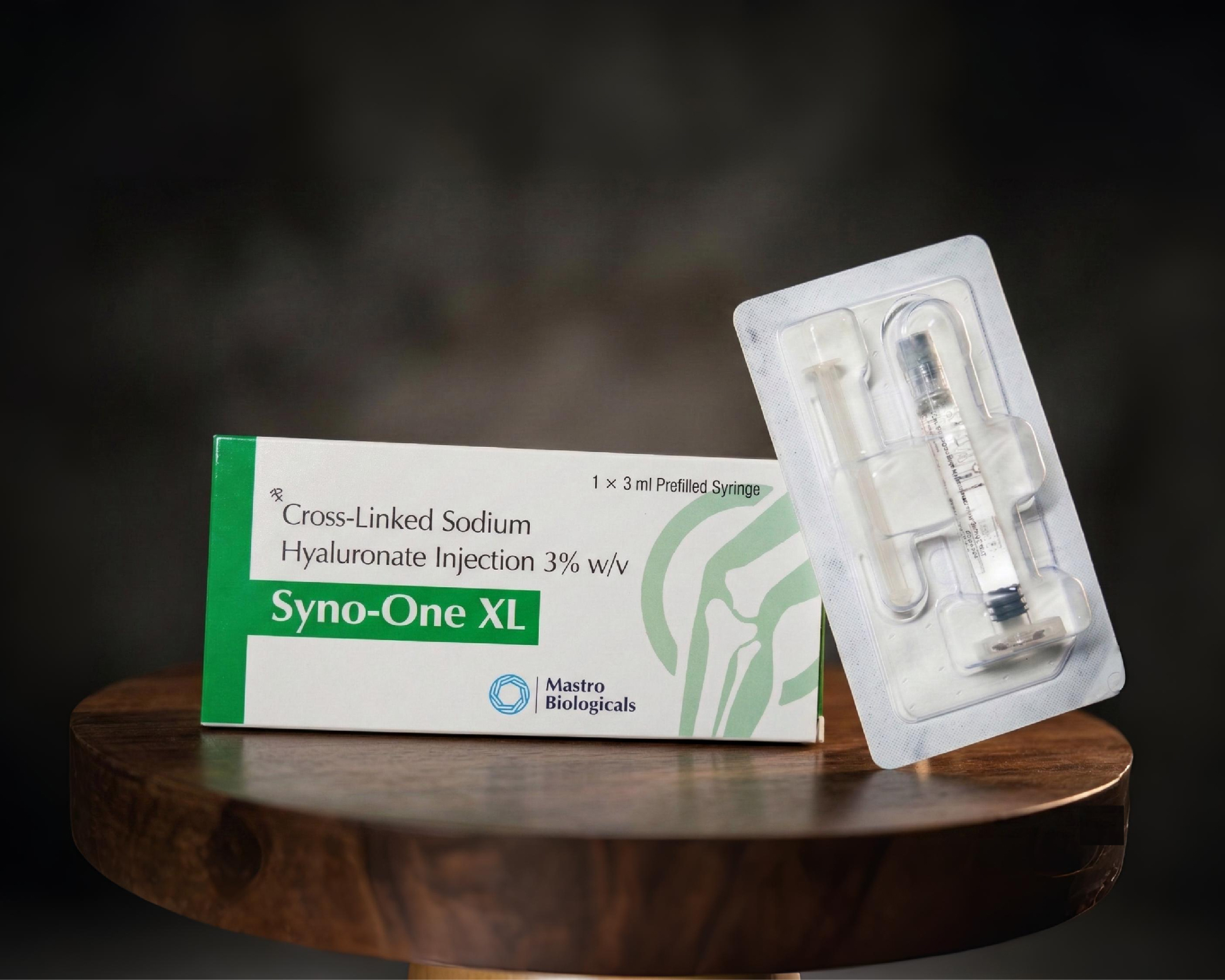
Syno one XL - Sodium Hyaluronate Injection
Class: Viscosupplement / Intra-articular lubricant
Molecule Type: Non-animal stabilized hyaluronic acid (NASHA)
Scientific Overview
Syno One contains high-molecular-weight sodium hyaluronate designed to restore synovial viscoelasticity in osteoarthritic joints. In OA, endogenous hyaluronic acid becomes depolymerised and loses its natural lubricating and shock-absorbing properties. Exogenous hyaluronate injections replenish viscosity, reduce synovial friction, and modulate intraarticular inflammation.
- Replenishes depleted synovial HA
- Forms a viscoelastic film over cartilage surfaces
- Reduces inflammatory cytokines
- Enhances boundary lubrication
- Improves biomechanical cushioning and shock absorption
- Pain Reduction: Significant reduction in VAS pain scores with improved WOMAC mobility indices
- Lasting Relief: Provides symptom relief lasting 4–6 months. Supports joint homeostasis and may delay need for arthroplasty.
- Broad Indications: Mild to moderate knee OA, hip/shoulder/ankle OA, post-traumatic chondral degeneration, sports-related chondropathy

TENDCART
TENDCART represents the next frontier in musculoskeletal therapy — a proprietary bioactive matrix formulation designed to stimulate cartilage regeneration and support tendon repair through a unique combination of growth factor analogues and structural scaffold components.
Currently in advanced clinical development, TENDCART is poised to address an unmet need in patients with cartilage defects and tendinopathies who are not yet candidates for surgical intervention but have exhausted conventional conservative options.
Osteoporosis & Bone Health
Advanced bone-protective therapies focused on:
- Increasing bone mineral density
- Reducing fracture risk
- Long-term skeletal strengthening
- Post-menopausal and age-related bone care
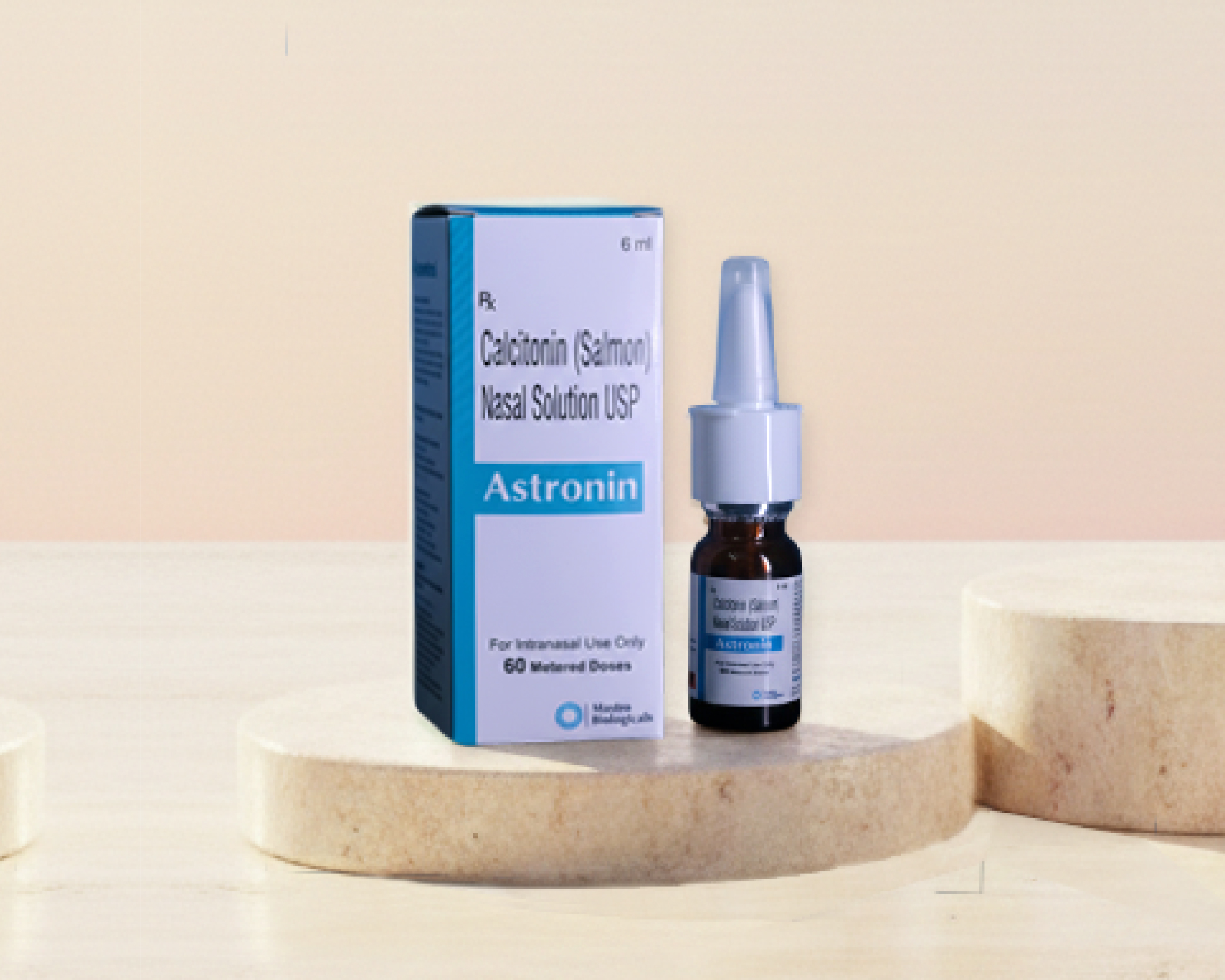
Astronin — Salmon Calcitonin
Anti-resorptive hormone that inhibits osteoclastic bone resorption. Stabilizes bone turnover, reduces acute vertebral fracture pain, and improves calcium homeostasis. For postmenopausal osteoporosis and vertebral compression fractures.
- Binds to osteoclast receptors
- Inhibits osteoclastic proton pump activity
- Reduces bone resorption surfaces
- Exhibits analgesic effect via endorphin modulation, useful in acute bone pain
- Postmenopausal osteoporosis
- Osteopenia transition therapy
- Pain from vertebral compression fractures
- Paget's disease of bone (supportive)
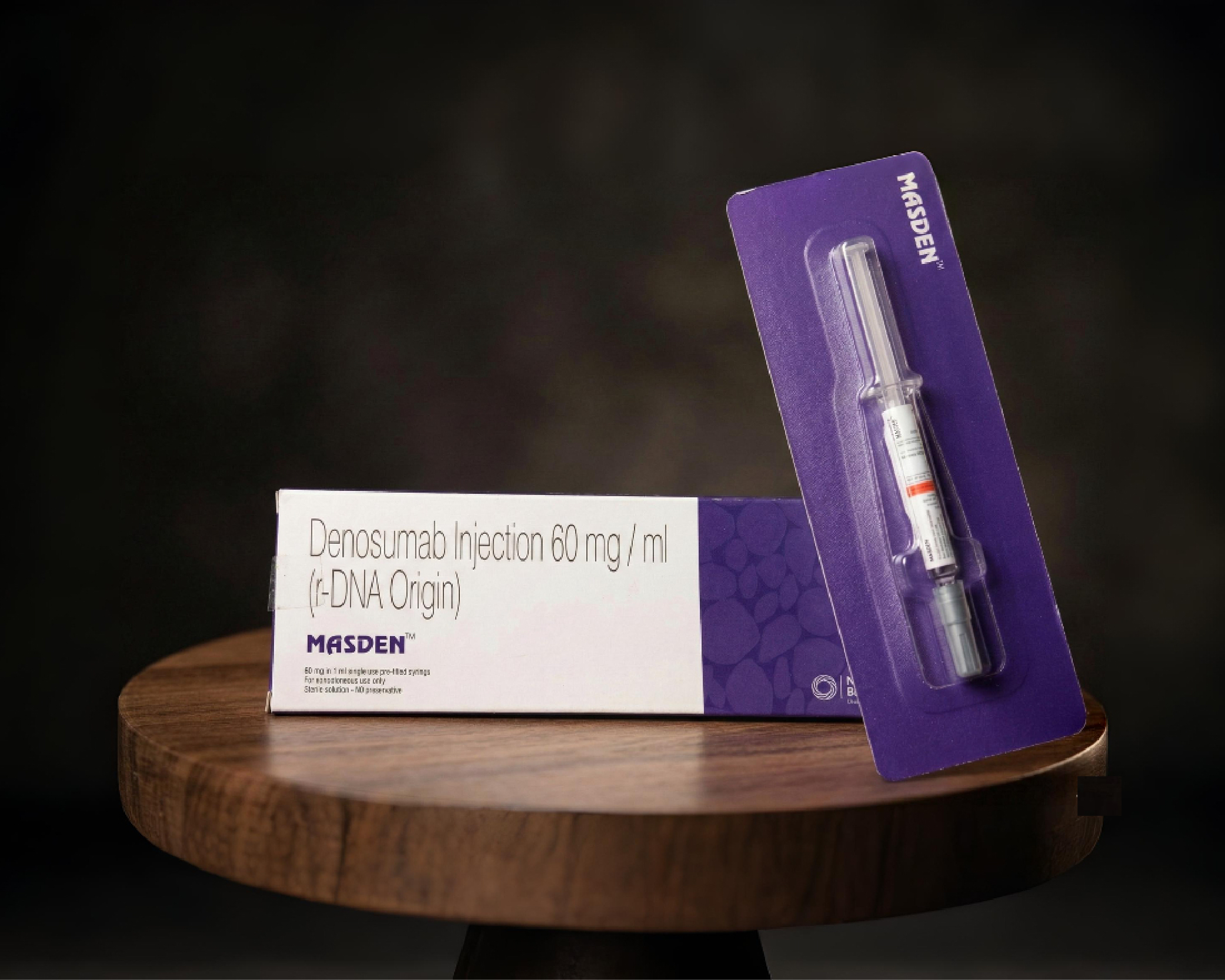
Masden — Denosumab
RANKL Inhibitor (IgG2). Blocks RANKL–RANK interaction, preventing osteoclast formation. Reduces vertebral fractures by up to 68% and hip fractures by 40% (FREEDOM study). No renal dose adjustments required.
Mechanism
Blocks RANKL–RANK interaction, preventing formation, function, and survival of osteoclasts. Decreases cortical & trabecular bone resorption. Increases BMD at hip, lumbar spine, and femoral neck. Indicated for postmenopausal osteoporosis, osteoporosis in men, steroid-induced osteoporosis, and high-fracture-risk patients.

Axiatide — Teriparatide
Anabolic bone-forming agent (PTH 1-34). Stimulates osteoblast proliferation and builds new bone. Increases lumbar spine BMD by 9–13%, reduces vertebral fractures by 65% and nonvertebral fractures by 53%.
Mechanism
Intermittent PTH (1-34) activates cAMP/PKA pathway, promotes osteoblast recruitment & reduces apoptosis. Enhances trabecular connectivity and bone microarchitecture. Ideal for severe osteoporosis with multiple fractures or ineffective response to antiresorptives. Also indicated for osteoporosis in men and GIOP.
TENDCART
TENDCART's bioactive matrix technology holds particular promise in osteoporosis management, where the compromised bone-cartilage interface presents unique therapeutic challenges. Its scaffold components are designed to support structural repair at the subchondral level — addressing the root cause of joint degeneration in osteoporotic bone.
By promoting integrated bone and cartilage repair, TENDCART aims to offer osteoporosis patients a treatment option that addresses joint degeneration at its structural source, complementing systemic antiresorptive therapies.
The Future of Bone-Cartilage Repair
Preclinical models of osteoporosis-related OA show TENDCART promoting mineralisation of the tidemark zone and restoration of the osteochondral unit — a unique mechanism of action with significant implications for osteoporosis-driven joint disease.
- Bioactive scaffold components support osteochondral repair by providing structural guidance for both chondrocyte and osteoblast lineage cells at the bone-cartilage interface.
- Growth factor analogues stimulate osteocalcin and collagen type II expression in preclinical models of osteoporosis-related subchondral bone loss.
- Potential to arrest the vicious cycle of subchondral bone deterioration and cartilage loss that characterises osteoporosis-related joint degeneration.
- Complementary mechanism to antiresorptive drugs; designed to work synergistically within existing osteoporosis treatment frameworks rather than replace them.
- Osteoporosis-associated OA is a designated sub-population within the TENDCART Phase III programme, with dedicated efficacy endpoints including DXA and MRI outcomes.
- Collaboration with leading bone metabolism research centres is underway to characterise osteochondral repair mechanisms in greater clinical detail.
- Regulatory fast-track designation is being pursued in key markets given the significant unmet need in osteoporosis-related joint disease management.
- Clinicians managing osteoporosis patients are encouraged to register interest in TENDCART clinical trial participation through the medical affairs contact portal.
Pain Management & Neuropathic Care
Evidence-based neuropathic pain solutions addressing peripheral neuropathy, chronic musculoskeletal pain, radiculopathy-associated symptoms, and functional pain restoration.

Masgaba-NT
Class: Neuropathic pain modulator / Co-analgesic
Mechanism of Action
Gabapentin: Reduces presynaptic calcium influx, decreases release of glutamate and substance P, dampens ectopic neuronal firing.
Nortriptyline: Enhances descending inhibitory pain circuits, exhibits antihyperalgesic activity, modulates central sensitization.
- Strong evidence for diabetic neuropathy
- Reduces allodynia & shooting neuropathic pain
- Better sleep quality and improved quality of life
- Effective in chronic radiculopathy and nerve compression
Syno One Plus
For patients requiring faster and more complete pain control, Syno One Plus combines the sustained pain-modulating properties of viscosupplementation with the rapid analgesic and anti-inflammatory action of a targeted corticosteroid component — providing immediate relief alongside long-term joint protection.
This formulation is ideally positioned within step-up pain management protocols, serving patients who have experienced partial but insufficient response to either single-agent analgesia or standard viscosupplementation.
Optimising Pain Outcomes
The complementary onset profiles of the two active components — immediate corticosteroid action within hours and progressive HA benefit over weeks — deliver a pain control curve uniquely suited to the variable, episodic nature of chronic joint pain.
- Corticosteroid component delivers clinically meaningful pain reduction within 24–48 hours of injection, providing immediate relief for patients with high acute pain burden.
- HA component produces progressive, cumulative pain reduction reaching peak effect at 4–8 weeks and sustained through 26 weeks in clinical trial populations.
- Combined VAS score reductions significantly exceed those achieved with either component alone, demonstrating additive analgesic synergy between the two active ingredients.
- Post-injection analgesic sparing allows clinicians to initiate planned dose reduction of systemic analgesics from week one, supporting safer long-term pain management.
- Optimal for patients with moderate-to-severe joint pain where rapid symptom control is clinically necessary — including pre-operative optimisation and post-exacerbation rescue therapy.
- Integration into structured pain management plans is recommended; injection should be coordinated with physiotherapy initiation to maximise functional rehabilitation gains.
- Minimum three-month retreatment interval; treatment plan review at six weeks recommended to assess analgesic response and plan subsequent pain management steps.
- Special consideration required in patients with adrenal suppression risk; full corticosteroid precaution protocol applies as per standard injectable steroid guidelines.
Syno One XL
Hip and shoulder pain are among the most functionally debilitating pain presentations in musculoskeletal practice. Syno One XL's extended-volume formulation ensures complete analgesic coverage of these large joint spaces, addressing pain arising from the full articular surface rather than only the area reached by standard injection volumes.
For patients with large-joint pain that has been refractory to oral and topical analgesics, Syno One XL offers a targeted, evidence-based intra-articular option with a compelling safety and efficacy profile.
Large Joint Pain: A Different Challenge
Undertreated hip and shoulder pain drives significant disability and opioid escalation. Syno One XL's adequate joint coverage directly addresses the inadequate distribution problem that limits analgesic outcomes with standard-volume formulations in large joints.
- Prospective data from hip OA cohorts shows mean VAS reduction of 42% at 12 weeks versus 24% with standard-volume HA, attributable to improved joint space coverage.
- Shoulder pain outcomes data demonstrate significant ASES and DASH score improvements at 12 and 26 weeks in patients with glenohumeral OA treated with Syno One XL.
- Opioid reduction of 35% at six months was documented in a real-world hip pain registry study of patients receiving repeat Syno One XL treatment cycles.
- Functional improvements in gait, range of motion, and daily activity scores correlate strongly with pain relief outcomes across all large joint indications studied.
- Imaging-guided administration is mandatory for hip injections; combination with post-injection physiotherapy produces superior pain and function outcomes versus injection alone.
- Suitable for integration into interdisciplinary pain clinic programmes as a non-opioid, non-systemic large joint analgesic option with a favourable safety and tolerability profile.
- Pain diary and patient-reported outcome measure tracking recommended from injection date to quantify individual analgesic response and inform retreatment timing decisions.
- Well tolerated in elderly patients and those with significant comorbidities where systemic analgesic options are limited by tolerability or interaction risk.
TENDCART
Unlike conventional analgesics that manage pain symptoms, TENDCART targets the structural tissue damage that drives persistent pain in cartilage and tendon disorders. By promoting genuine tissue repair, TENDCART aims to offer durable pain relief that reduces and potentially eliminates the need for ongoing symptomatic treatments.
This disease-modifying approach to pain management represents a paradigm shift — addressing the underlying pathology rather than masking its symptoms, with early data suggesting sustained pain reduction well beyond treatment completion.
The Disease-Modifying Advantage
For patients trapped in cycles of escalating analgesic use, TENDCART's regenerative mechanism offers a credible exit pathway — restoring tissue integrity and eliminating the structural pain source rather than suppressing the pain signal alone.
- Tissue regeneration reduces the structural pain generators (cartilage erosion, tendon micro-tears) that drive persistent nociceptor activation in chronic musculoskeletal pain.
- Early data suggests pain reduction scores continue to improve after the acute treatment phase, consistent with the progressive nature of tissue repair rather than symptom suppression.
- Potential to break the chronification cycle of structural damage → inflammation → sensitisation → further damage that characterises progressive joint and tendon pain disorders.
- Anti-inflammatory bioactive components concurrently suppress the inflammatory mediators responsible for peripheral sensitisation and central pain amplification.
- Chronic knee pain from cartilage defects, refractory Achilles tendinopathy, and persistent patellar tendon pain are the primary pain indications in the TENDCART Phase III programme.
- Pain outcomes assessed using NRS, KOOS, VISA-A, and PROMIS Pain Interference scales to comprehensively characterise the analgesic benefit profile across indications.
- Combination with structured pain rehabilitation programmes is planned in trial arms to evaluate the additive benefit of TENDCART within multimodal pain pathways.
- Pain management specialists and rheumatologists interested in the TENDCART early access programme should contact the medical affairs team for eligibility information.
Autoimmune & Rheumatology
Targeted immunomodulatory therapies for precision-driven inflammatory joint management. Disease-modifying approach for rheumatoid arthritis with long-term inflammation
MJAK-T
Class: JAK Inhibitor (JAK1 & JAK3 predominant) | Oral targeted synthetic DMARD(tsDMARD)
Mechanism
Inhibits JAK1/JAK3, suppresses STAT dependent transcription. Reduces IL-2, IL-4, IL-6, IL-7, IL-15, IL-21 signalling. Decreases T-cell activation and rapidly reduces synovial inflammation.
- Reduces RA symptoms as early as Week 2. Improves HAQ-DI and ACR response rates. Oral therapy — no injections needed. Prevents long-term joint erosions.
- Rheumatoid arthritis (moderate to severe), psoriatic arthritis, autoimmune polyarthropathy, and inadequate responders to MTX or biologics.
Syno One Plus
Autoimmune joint flares are a defining feature of conditions like RA and PsA, causing acute inflammation that erodes joint integrity and severely impacts patient quality of life. Syno One Plus delivers targeted intra-articular corticosteroid therapy for flare control alongside lasting HA-mediated joint protection — a clinically powerful combination in autoimmune arthropathy management.
Particularly valuable as a bridge therapy during biologic treatment transitions or step-up DMARD changes, Syno One Plus controls localised joint inflammation while systemic agents reach therapeutic effect.
Bridging Therapy in Autoimmune Disease
When systemic therapy changes leave patients temporarily under-protected, Syno One Plus provides reliable local flare control and joint preservation — maintaining patient function and wellbeing through the treatment transition window.
- Intra-articular corticosteroid component provides rapid, targeted suppression of localised synovitis with substantially lower systemic immunosuppressive burden than equivalent oral corticosteroid doses.
- HA component protects articular cartilage from the erosive effects of inflammatory enzymes released during flares, contributing to long-term joint preservation goals.
- Validated for use in RA and PsA flares; clinical response rates comparable to systemic corticosteroid rescue therapy with a more favourable systemic safety profile.
- Reducing oral steroid use through targeted intra-articular administration supports osteoporosis prevention objectives in patients receiving long-term immunosuppressive regimens.
- Heightened infection risk in immunosuppressed autoimmune patients requires strict aseptic technique; joint aspiration and synovial fluid culture should be considered to exclude septic arthritis before injection.
- Cumulative corticosteroid burden tracking is essential in autoimmune patients receiving multiple intra-articular injections alongside systemic DMARDs and biologics.
- Three-month minimum retreatment interval; coordinate injection schedule with rheumatology team to align with broader disease management and treatment review cycles.
- Patient counselling on infection awareness post-injection is mandatory given elevated baseline immunosuppression from concurrent biologic and/or DMARD therapy.
Syno One XL
Systemic autoimmune conditions such as RA and AS frequently affect large joints including the hip, shoulder, and sacroiliac joint, where standard injection volumes are inadequate to provide full therapeutic coverage. Syno One XL ensures complete synovial treatment of these larger affected joint compartments.
For autoimmune patients with large-joint involvement refractory to systemic therapy alone, Syno One XL provides a targeted intra-articular option that complements biologic treatment without adding systemic immunosuppressive burden.
Large Joint Autoimmune Disease
In conditions like AS and RA where hip involvement drives significant disability, Syno One XL's validated large-joint efficacy offers a meaningful addition to treatment strategies that may otherwise have limited intra-articular options.
- Clinical data supports use in RA-related hip and shoulder arthropathy, with WOMAC and HOOS improvements at 12 weeks consistent with those observed in primary OA populations.
- Adequate volume coverage of the hip joint space — achieved only with the XL formulation — correlates with superior HA retention and more durable analgesic outcomes in large-joint disease.
- Particularly valuable in AS patients with hip involvement where systemic biologic therapy controls spinal disease but residual hip pain and stiffness persist.
- Improves functional mobility metrics relevant to independence and quality of life in autoimmune patients managing complex, multi-joint disease burdens.
- Imaging guidance mandatory for all hip administrations; ultrasound or fluoroscopy confirmation of intra-articular placement is essential for safety and efficacy in this patient population.
- Infection screening and clinical examination to exclude active synovitis or septic arthritis should precede every injection in immunosuppressed autoimmune patients.
- Integration into the broader rheumatology treatment plan is recommended; coordinate injection timing with DAS28 assessments and biologic administration schedules to optimise outcomes.
- Retreatment at six-month intervals supported; long-term use alongside stable biologic therapy has been observed to maintain functional improvements without cumulative safety concerns.
TENDCART
Autoimmune arthropathies are characterised by progressive, immune-mediated cartilage and connective tissue destruction that current systemic therapies slow but cannot reverse. TENDCART's regenerative bioactive matrix technology is specifically designed to address this irreversible structural damage — the critical unmet need in long-term autoimmune joint management.
By combining structural repair capability with localised anti-inflammatory activity, TENDCART could offer autoimmune patients the first genuine opportunity to recover, rather than merely preserve, joint function in the biologic era.
Repairing Autoimmune Joint Damage
Even with optimal systemic disease control, cartilage lost to years of autoimmune inflammation cannot be recovered by current therapies. TENDCART's regenerative mechanism opens a new therapeutic dimension in autoimmune joint care.
- Bioactive scaffold provides structural guidance for chondrocyte and synoviocyte repopulation in joints where autoimmune inflammation has created significant tissue voids.
- Localised anti-inflammatory matrix components suppress residual MMP and RANKL activity within the joint, protecting newly regenerated tissue from immune-mediated re-destruction.
- Designed to function within the immunosuppressed joint environment created by biologic therapy, leveraging systemic disease control to optimise the regenerative microenvironment.
- Preclinical data in collagen-induced arthritis models demonstrates cartilage matrix deposition and GAG content recovery, supporting the feasibility of repair even in autoimmune joint environments.
- RA-related cartilage defects and PsA joint destruction are designated autoimmune sub-populations within the TENDCART Phase III programme, with separate efficacy endpoints from primary OA arms.
- Safety in immunosuppressed patients receiving biologics and DMARDs is a primary safety endpoint; current Phase II data shows no increase in injection-site infection rates versus control.
- Immunogenicity profiling of all TENDCART components has been completed; no clinically significant immune reactions to scaffold or growth factor components have been identified in Phase II.
- Rheumatologists interested in enrolling autoimmune patients in the TENDCART trial are invited to contact the clinical development team for site qualification and protocol details.
